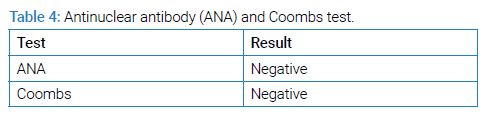Abstract
Sickle cell disease is a well-known cause of hemolytic anemia, which causes several systemic complications. Cerebral Fat Embolism (CFE) secondary to avascular necrosis of the bones is a rare, known complication of sickle cell disease. Here, we report a case of a young gentleman who presented with an acute onset of progressive drowsiness, followed by fever, generalized body aches, and shortness of breath secondary to CFE. CFE usually happens secondary to the dislodgement of fat particles from within the body, either secondary to fracture of a long bone or instrumentation of an atherosclerotic vessel or as a complication of medical illness (for example, sickle cell disease). Fat particles can become dislodged in a variety of organs involving the brain, lungs, spleen, kidney, etc., resulting in widespread systemic manifestations (altered mentation, hypoxia, splenomegaly, abdominal tenderness, acute kidney injury, and petechial rashes). As mentioned earlier, CFE due to avascular necrosis in sickle cell disease is a known yet rare complication. This case would add to the limited literature on this topic, providing awareness regarding sickle cell disease in patients presenting with CFE. This would help in early and prompt diagnosis, guide treatment plans, avoid unnecessary procedures and investigation, and ultimately improve patient outcomes.
Case Presentation
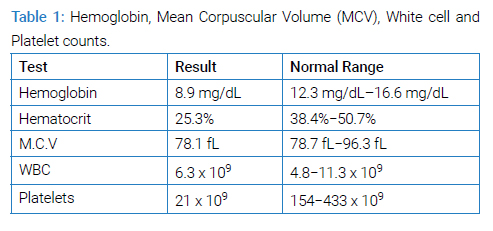
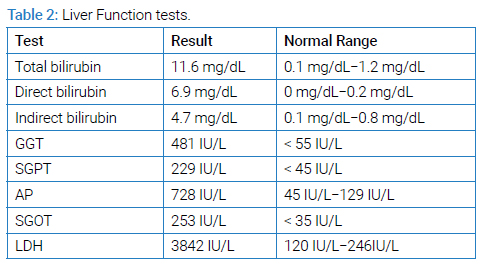
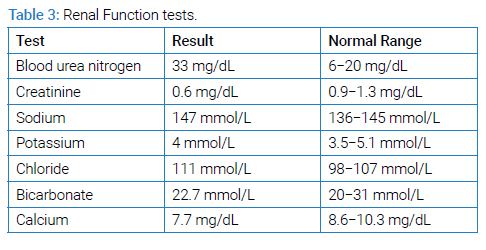
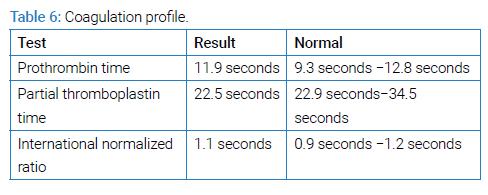
A young male of 30 years of age with a known case of sickle cell disease presented with complaints of fever, generalized body ache, and shortness of breath for two weeks. There was no associated orthopnea or paroxysmal nocturnal dyspnea. No history of regular blood transfusions or phlebotomies except one session of phlebotomy during his recent illness. He was initially hospitalized in another hospital, where he was managed along the lines of hemolytic anemia and pneumonia. During the hospital stay, he developed an altered mental status, so he was brought to our hospital for further management. On arrival at the emergency department, he had a Glasgow Coma Scale (GCS) of 9/15 (E2 M5 V2). A limited neurological examination showed pupils of 2 mm bilaterally equally reactive to light and increased tone throughout the body with mute planters bilaterally. The cough and Gag reflexes were intact. He was hypoxic, with an oxygen requirement of up to 5 liters with a face mask. Petechial rashes were noted on the chest and bilateral forearms. He was admitted to the intensive care unit. After initial resuscitation, blood workup including complete blood count parameters (Table 1), liver function test (Table 2), renal function test and electrolytes (Table 3) ANA and Coomb’s test (Table 4), hemoglobin electrophoresis (Table 5) and coagulation profile (Table 6) was done. The complete blood count was consistent with microcytic anemia. White blood cell count was within normal range, which depicted the non-infectious etiology of his symptoms. A Liver Function Test (LFT) was performed, and the total bilirubin was raised with increased direct and indirect bilirubin. A raised LDH and increased bilirubin favored the diagnosis of hemolysis. Coagulation studies were within normal ranges. Results of Hb electrophoresis showed a high percentage of Hb S was raised to establish the diagnosis of sickle cell disease. Coagulation studies were done as a part of the initial workup, which was normal.
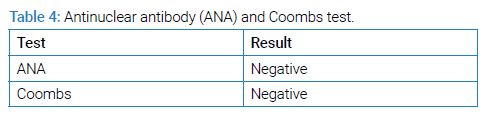

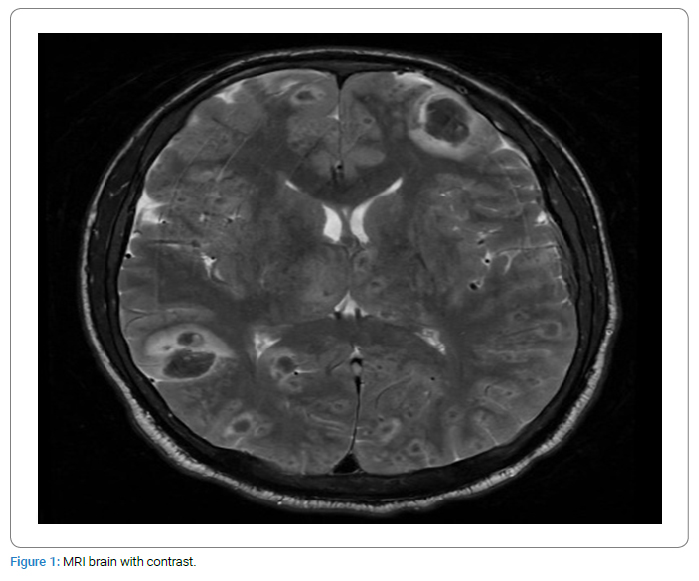
MRI brain with contrast (Figure 1) was done which showed widespread multifocal areas of signal abnormality in both supra and infra-tentorial compartment of the brain, involving both grey and white matter. These areas returned iso-intense signals on T1-weighted images hypo-intense signals on T2-weighted images, and signal dropouts on SWI images represented hemorrhages with associated mild vasogenic oedema. Findings were suggestive of widespread fat embolism with multiple micro-hemorrhages.
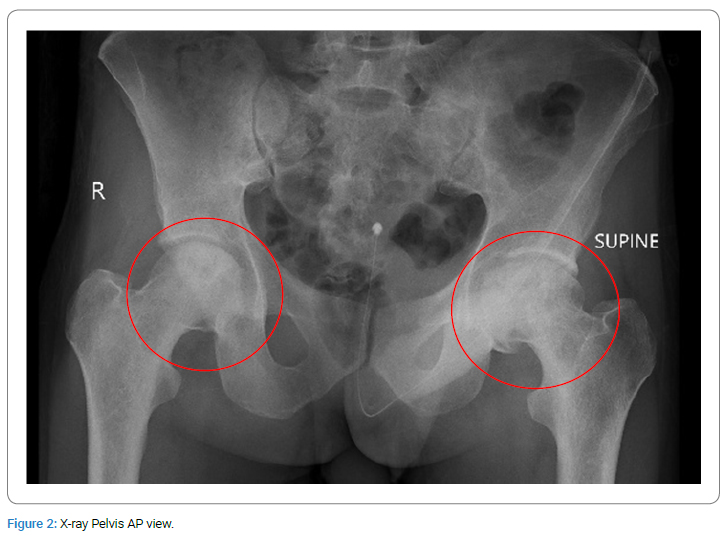
An X-ray of the pelvis was performed to look for avascular necrosis of the head of the femur. It is fairly common in sickle cell disease. As much as 50% of sickle cell patients can develop AVN by the time they reach the age of 35 [1].
The pelvic X-ray (Figure 2) revealed (in red circles) flattening of the left femoral head with patchy sclerosis and lucency, representing avascular necrosis. Patchy sclerosis and lucency were also identified in the right femoral head without any contour abnormality representing avascular necrosis.
CT chest performed earlier at another hospital for persistent hypoxia revealed interlobular septal thickening with superimposed ground glass opacities. These findings raise concerns about bilateral alveolar hemorrhages. However, there was no obvious history of hemoptysis.
The gentleman was managed conservatively with intravenous fluids, broad-spectrum intravenous antibiotics, intravenous analgesia, and intravenous antiepileptic. Supplemental oxygen was administered. He was planning to undergo further blood workup and treatment, but his family had severe financial constraints. Hence, the left went against medical advice within one day.
Discussion
Sickle cell disease is an inherited disorder affecting the red blood cell morphology and its oxygen-carrying capacity, causing hemolysis and a variety of systemic manifestations. Sickle cell crisis is an acute presentation of sickle cell disease. A sickle crisis is comprised of uncontrolled sickling of RBCs, resulting in hemolytic and vaso-oclusive effects. Hemolysis results in features of anemia, fatigue and even raised bilirubin. On the other hand, vaso-occlusion causes shortness of breath, chest pain, headache, cough, and bone pains, all due to reduced blood flow, resulting in tissue starvation. It is important to remember that conditions of dehydration, infection, and even certain medications can result in uncontrolled sickling in patients with sickle cell disease. Vaso-occlusion can cause various complications, including recurrent bacterial infections, stroke, cholelithiasis, priapism, osteomyelitis, acute papillary necrosis, leg ulcers, etc. [2]. The management of crisis episodes depends upon the organ involved in addition to hydration, analgesia, and supplemental oxygen [7]. A rare and serious complication is cerebral fat embolism (CFE), which is a rare yet serious known complication of sickle cell disease. Information on CFE in sickle cell disease is limited, and it can have devastating consequences [3]. According to research conducted, fat embolism can happen in as low as 1% of traumatic or nontraumatic cases [4]. It is mostly the result of the dislodgement of fat particles from the marrow of long bones or after the intervention in an atherosclerotic vessel with resultant CNS, dermatological, i.e., petechial rash, and hematological symptoms, i.e., thrombocytopenia. The triad of drowsiness, petechial skin rash, and low platelets as a consequence of fat embolism is called Fat Embolism Syndrome (FES) [3]. The percentage of CNS involvement in FES is around 59% [5]. It can initially present with vaso-occlusive crises, with later development of CFE [6]. Symptomatology depends upon the organ system involved, which can range from skin rash, shortness of breath, abdominal pain, decreased level of consciousness, dropping urine output, and fever [7,8].
In the context of sickle cell disease, very little information is available about the CFE happens secondary to bilateral avascular necrosis of the bilateral femoral heads [9–11]. Development of the altered sensorium, focal neurological deficits, and mute plantar responses can be useful signs and symptoms of CFE, along with a typical rash on the body. There can be the development of fragile collaterals in the brain secondary to the occlusive vasculopathy, giving rise to a syndrome known as the Moya-moya syndrome [12]. Brain imaging plays a pivotal role in the exclusion of other causes of CNS pathology, with the demonstration of multiple cavitary lesions with hyperintense signals with surrounding vasogenic edema on the MRI brain [13].
In many instances, the diagnosis of fat embolism syndrome is made during autopsy because of the non-availability of any sensitive or specific test for the diagnosis of CFE [14]. Studies have shown that certain scoring systems like the Gurd and Wilson criteria and the Schonfeld Fat.
Embolism Index can play a role in identifying patients with FES [14]. The role of neuroimaging in this regard is very promising, and can show characteristic findings on susceptibility weighted imaging [15].
There are no specific treatments available for CFE, but studies have shown the role of red cell exchange transfusion and therapeutic plasma exchange as lifesaving measures [16]. Mortality was reported to be as low as 29% in those sickle cell disease patients who were treated with exchange transfusion, in comparison to those who were not given exchange transfusion (91%) [17]. Hematopoietic stem cell transplant can be done with the intent of cure in patients with sickle cell disease [18]. Apart from this, supplemental oxygen and non-invasive ventilation are needed to relieve respiratory distress and hypoxia. The role of extracorporeal membrane oxygenation in the treatment of pulmonary embolism is well documented [19]. Worsening consciousness level with poor respiratory efforts warrants mechanical ventilation. Pain control with the help of strong analgesics [20] and frequent GCS monitoring is required [21].
Conclusion
Cerebral fat embolism can have lethal outcomes in patients with Sickle cell disease. Acute development of altered mental status in a patient known to have sickle disease should prompt us to look for cerebral fat embolism by brain imaging. MRI brain can be helpful in diagnosing this lethal complication of sickle cell disease and can show peculiar findings suggestive of cerebral fat embolism. Early initiation of exchange transfusion can be a lifesaving therapeutic measure.
Conflict of Interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Informed consent was obtained for this publication.
Keywords
Sickle cell anemia; Fat embolism; Avascular necrosis
Cite this article
Wasti DR, Hanif DR. From blood to bone and brain: case report of fat embolism as initial presentation of sickle cell disease. Clin Oncol J. 2024;5(1):1–5.
Copyright
© 2024 Wasti DR. This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY-4.0).